Definition and structure of Protein: Proteins may be defined as the high molecular wright mixed polymer of alpha-amino acids joined together with peptide bond (-CO-NH-). Polymers of more thane 100 amino acids (usually have a molecular weight above 10000) constituents of all living matter.
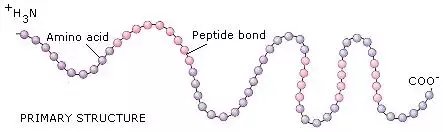
Primary structure of protein
The alpha-amino acid units are linked together through the carboxyl and amino groups to produce the primary structure of the protein chain. The bond between two adjacent alpha-amino acids known as the peptide bond is in fact the backbone of the protein chain.

Secondary structure of protein
A coiled structure of protein in which peptide bonds are folded in a regular manner. The bondings are the result of the linking of the alpha-carboxyl and alpha-amino groups of the peptide chains by means of hydrogen bonds and disulfide bonds. Such bondings are referred to as the secondary structure of the protein.
Classification of protein
Protein can be classified in two ways- based on solubility and based on function.
Classification of protein based on solubility
Simple proteins are classified on their solubility basis as follows-
1. Albumins: these are soluble in water; precipitated with a saturated solution of salt like ammonium sulphate. e.g. serum albumin, egg albumin, lactalbumin(milk) legumelin (soyabeans)
2. Globulin: these are insoluble in water but soluble in dilute salt solution, precipitated with half saturated solution of ammonium sulphate. e.g. serum globulin, vitellin (egg yolk), legumin(peas)
3. Glutelins: These are insoluble in water but soluble in dilute acids and alkalis. e.g. Glutenin (wheat), Oryzenin (rice)
4. Prolamin: These are insoluble in water and absolute alcohol but soluble in 70 to 80% alcohol. e.g. Gliadin (wheat), zein (maize)
5. Protamine: these are soluble in water dilute acids and alkalis. e.g. salmine, salmon sperm.
6. Histones: these are soluble in water and insoluble in every dilute ammonium hydrogen. e.g. Globin of haemoglobin.
7. Seleroprotein: These are insoluble in water, dilute solutions of acids, bases, and salts. e.g. Collagen(bone, skin) , elastin (ligament), keratin (hair, nail).
Classification of protein based on function
Classification of protein according to their function is given below-
1. Transport Protein: Transport protein in blood plasma bind and carry specific molecules or ions from one organ to another. For example, Haemoglobin of blood bind oxygen and release oxygen to cells for energy -yielding oxidation of nutrients. The blood plasma contains lipoproteins, which carry lipids from the liver to other organs.
2. Nutrients and storage proteins: The seeds of many plants store nutrient proteins required for the growth of the germinating seedling.
For example: legumin (storage protein of legume) seeds, Glutenins (storage protein of wheat seed) gliadins (storage protein of wheat seed) Glutelins (storage protein of rice).
In addition to above seed storage protein, ovalbumin, the major protein of egg white, and casein, the major protein of milk, are other exam less of nutrient proteins.
3. Contractile or motile proteins: some protein endow cells with the ability to contract or to move about. Actin and myosin function in the contractile system of skeletal muscle.
4. Structural proteins: Many proteins give biological structures strength or protection. The major component of bones and cartilage is the protein, collagen. Other examples, are Ligaments contain the protein, elastin, hair and. Fingernails consists largely of the protein, keratin.
5. Defence proteins: Many proteins defend organisms against invasion by other species or protect theme from injury. The antibodies (Proteins) precipitate or neutralize invading bacteria, viruses or other microorganisms. Fibrinogen and thrombin are blood coating proteins that prevent loss of blood from injury.
6. Regulatory proteins: some proteins help to regulate Cellular or Physical activities. Among them are many hormones. Examples: insulin hormone (also protein) regulates sugar metabolism and the growth hormone of the pituitary
7. Other proteins: Monellin, a protein of an African Plant, has an intensely sweet taste. The blood plasma of some Antarctic fish contains antifreeze protein.